|
Is ketosis dangerous? If so, your doctor is confusing diabetic ketoacidosis (DKA) with nutritional ketosis, or keto- adaptation. For starters, it’s a vital evolutionary advantage. When a diabetic (usually a Type I diabetic, but sometimes this occurs in very late- stage, insulin- dependent, Type II diabetics) fails to receive enough insulin, they go into an effective state of starvation. Background Long-term microvascular and neurologic complications cause major morbidity and mortality in patients with insulin-dependent diabetes mellitus (IDDM). Insulin resistance is a condition that is a precursor to developing type 2 diabetes. Causes of insulin resistance include metabolic syndrome, pregnancy, stress. Information about insulin reaction (excessively low blood sugar levels) from exercise, skipped meals, or medication errors. Symptoms include sweating, shaking. Is there a special diet plan for treating insulin resistance? What foods help prevent type 2 diabetes? What about exercise to treat insulin resistance? 
Insulin resistance - Wikipedia. Insulin resistance (IR) is a pathological condition in which cells fail to respond normally to the hormone insulin. The body produces insulin when glucose starts to be released into the bloodstream from the digestion of carbohydrates in the diet. Normally this insulin response triggers glucose being taken into body cells, to be used for energy, and inhibits the body from using fat for energy. The concentration of glucose in the blood decreases as a result, staying within the normal range even when a large amount of carbohydrates is consumed. When the body produces insulin under conditions of insulin resistance, the cells are resistant to the insulin and are unable to use it as effectively, leading to high blood sugar. Beta cells in the pancreas subsequently increase their production of insulin, further contributing to a high blood insulin level. 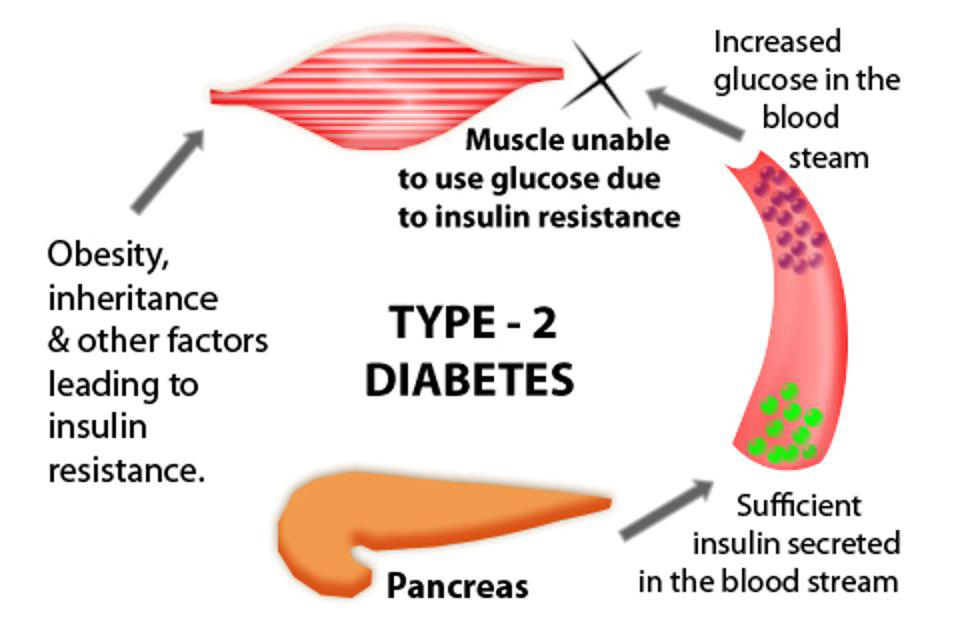 
This often remains undetected and can contribute to the development of type 2 diabetes or latent autoimmune diabetes of adults. Recent investigations have revealed that insulin resistance helps to conserve the brain's glucose supply by preventing muscles from taking up excessive glucose. Insulin resistance is a syndrome (a set of signs and symptoms) resulting from reduced insulin activity; it is also part of a larger constellation of symptoms called the metabolic syndrome. Insulin resistance may also develop in patients who have recently experienced abdominal or bariatric procedures. At the molecular level, a cell senses insulin through insulin receptors, with the signal propagating through a cascade of molecules collectively known as PI3. K/Akt/m. TOR signaling pathway. From a broader perspective, however, sensitivity tuning (including sensitivity reduction) is a common practice for an organism to adapt to the changing environment or metabolic conditions. This can be achieved through raising the response threshold (i. IRS) and PI3. K, which is the essence of the so- called adjustable threshold hypothesis of insulin resistance. Studies on animals have observed significant insulin resistance in rats after just 3 weeks on a high- fat diet (5. In humans, statistical evidence is more equivocal. Being insensitive to insulin is still positively correlated with fat intake, and negatively correlated with dietary fiber intake. The effect is thought to explain relatively low incidence of IR, type 2 diabetes, and obesity in polar foragers such as Alaskan Eskimos consuming their ancestral diet (which is very high in fat, but contains substantial amounts of omega- 3). Unlike their omega- 6 counterparts (which may be produced cheaply from a variety of sources, such as corn and soybeans), major sources of omega- 3 fatty acids remain relatively rare and expensive. Consequently, the recommended average intake of omega- 3 for adult men in the United States is only 1. 
Insulin resistance (IR) is a pathological condition in which cells fail to respond normally to the hormone insulin. The body produces insulin when glucose starts to. Insulin kits recalled for possibly fatal flaw 10/4/2016 - If you suffer from a serious diabetic condition, and rely on a medical injection device for insulin, beware. The metabolic abnormalities of non-insulin-dependent diabetes mellitus (NIDDM) are generally acknowledged to result from a combination of insulin resistance and. 
United States is approximately 1. ALA; EPA and DHA contributed less than 0. They are correlated with excess body weight. High- carbohydrate, low- fat diets were found by many studies to result in elevated triglycerides. Therefore, it may contribute to insulin resistance through the same mechanisms as dietary fat. Just like fat, high levels of fructose and/or sucrose induce insulin resistance in rats. In particular, very little is known about the effects of simple sugars in whole fruits and vegetables. If anything, epidemiological studies suggest that their high consumption is associated with somewhat lower risk of IR and/or metabolic syndrome. Leptin is a hormone that regulates long- term energy balance in many mammals. An important role of leptin is long- term inhibition of appetite in response to formation of body fat. This mechanism is known to be disrupted in many obese individuals: even though their leptin levels are commonly elevated, this does not result in reduction of appetite and caloric intake. This serves as a major motivation behind the low- carb family of diets. Furthermore, carbohydrates are not equally absorbed (for example, the blood glucose level response to a fixed quantity of carbohydrates in baked potatoes is about twice the response to the same quantity of carbohydrates in pumpernickel bread). Integrated blood glucose response to a fixed quantity of carbohydrates in a meal is known as glycemic index (GI). Some diets are based on this concept, assuming that consumption of low- GI food is less likely to result in insulin resistance and obesity, however, small to moderate amounts of simple sugars (i. 
Since insulin is the primary hormonal signal for energy storage into fat cells, which tend to retain their sensitivity in the face of hepatic and skeletal muscle resistance, IR stimulates the formation of new fatty tissue and accelerates weight gain. Some scholars go as far as to claim that neither insulin resistance, nor obesity really are metabolic disorders per se, but simply adaptive responses to sustained caloric surplus, intended to protect bodily organs from lipotoxicity (unsafe levels of lipids in the bloodstream and tissues): . Consumption of fast food has been proposed as a fundamental factor behind the metabolic syndrome epidemic and all its constituents. Cortisol counteracts insulin, contributes to hyperglycemia- causing hepatic gluconeogenesis. 
Mice without JNK1- signaling do not develop insulin resistance under dietary conditions that normally produce it. As short- term overdosing of insulin causes short- term insulin resistance, it has been hypothesized that chronic high dosing contributes to more permanent insulin resistance. This link seems to exist under diverse causes of insulin resistance. It also is based on the finding that insulin resistance may be reversed rapidly by exposing cells to mitochondrial uncouplers, electron transport chain inhibitors, or mitochondrial superoxide dismutase mimetics. It has long been observed that patients who have had some kinds of bariatric surgery have increased insulin sensitivity and even remission of type 2 diabetes. It was discovered that diabetic/insulin resistant non- obese rats whose duodenum has been removed surgically, also experienced increased insulin sensitivity. This suggested similar surgery in humans, and early reports in prominent medical journals. The speculation is, that some substance is produced in the mucosa of that initial portion of the small intestine that signals body cells to become insulin resistant. If the producing tissue is removed, the signal ceases and body cells revert to normal insulin sensitivity. No such substance has been found as yet, and the existence of such a substance remains speculative. Thus, glucose, fatty acids and amino acids 'leak' out of the cells. A decrease in insulin/glucagon ratio inhibits glycolysis which in turn decreases energy production. The resulting increase in blood glucose may raise levels outside the normal range and cause adverse health effects, depending on dietary conditions. When these cells fail to respond adequately to circulating insulin, blood glucose levels rise. The liver helps regulate glucose levels by reducing its secretion of glucose in the presence of insulin. This normal reduction in the liver's glucose production may not occur in people with insulin resistance. Insulin resistance normally refers to reduced glucose- lowering effects of insulin. However, other functions of insulin can also be affected.  
For example, insulin resistance in fat cells reduces the normal effects of insulin on lipids and results in reduced uptake of circulating lipids and increased hydrolysis of stored triglycerides. Increased mobilization of stored lipids in these cells elevates free fatty acids in the blood plasma. Elevated blood fatty- acid concentrations (associated with insulin resistance and diabetes mellitus Type 2), reduced muscle glucose uptake, and increased liver glucose production all contribute to elevated blood glucose levels. High plasma levels of insulin and glucose due to insulin resistance are a major component of the metabolic syndrome. If insulin resistance exists, more insulin needs to be secreted by the pancreas. If this compensatory increase does not occur, blood glucose concentrations increase and type 2 diabetes or latent autoimmune diabetes of adults occurs. In normal metabolism, the elevated blood glucose level instructs beta (. The insulin, in turn, makes insulin- sensitive tissues in the body (primarily skeletal muscle cells, adipose tissue, and liver) absorb glucose, and thereby lower the blood glucose level. The beta cells reduce insulin output as the blood glucose level falls, allowing blood glucose to settle at a constant of approximately 5 mmol/L (m. M) (9. 0 mg/d. L). In an insulin- resistant person, normal levels of insulin do not have the same effect in controlling blood glucose levels. During the compensated phase on insulin resistance, insulin levels are higher, and blood glucose levels are still maintained. If compensatory insulin secretion fails, then either fasting (impaired fasting glucose) or postprandial (impaired glucose tolerance) glucose concentrations increase. Eventually, type 2 diabetes or latent autoimmune diabetes occurs when glucose levels become higher throughout the day as the resistance increases and compensatory insulin secretion fails. The elevated insulin levels also have additional effects (see insulin) that cause further abnormal biological effects throughout the body. Insulin resistance often progresses to full Type 2 diabetes mellitus (T2. DM) or latent autoimmune diabetes of adults. The inability of the . Examples include infection (mediated by the cytokine TNF. Recent research is investigating the roles of adipokines (the cytokines produced by adipose tissue) in insulin resistance. Certain drugs also may be associated with insulin resistance (e. Exercise reverses this process in muscle tissue. With respect to visceral adiposity, a great deal of evidence suggests two strong links with insulin resistance.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
October 2017
Categories |
 RSS Feed
RSS Feed
